* Purpose of preclinical trials – Study the safety, effectiveness, and toxicity of test material “CM-7919 (As4O6)”
* Laboratories & Institutes – Biotoxtech, Korea Cancer Center Hospital, China A medical research institute
* Period of preclinical trials – 2001 ~ 2007, 2014 ~ 2016
ANTI –
ANGIOGENESIS
CONTENTS OF STUDY >
- To identify anti-angiogenesis effect and mechanism using endothelial cells
- To identify systemic anti-angiogenesis effect using rats
RESULTS >
- Identified inhibition of proliferation, migration, infiltration, and angiogenesis through in vitro study.
- Identified significant inhibition of systemic angiogenesis in in vivo experiment
- No death, weight loss, evacuation disorder, motor disorder, and abnormal behavior of experimental animals were observed
APOTOSIS
CONTENTS OF STUDY >
- To identify mechanism and effect of apoptosis of cancer cells
RESULTS >
- Identified mechanism and effect of apoptosis of cancer cells
- Confirmed apoptotic function of cancer cells including blood cancer, liver cancer, and bladder cancer.
TOXICITY
CONTENTS OF STUDY >
- Toxicity of dose once a day
- Toxicity of repetitive dose for 13 weeks
- Toxicity of repetitive dose for 26 weeks
- Toxicity of repetitive dose for 52 week
- Genetic toxicity (chromosome abnormality, mutation, micronucleus test)
RESULTS >
- Almost no adverse toxicity effect from single and repetitive dose
- No chromosomal abnormality
PHARMACOKINETICS
CONTENTS OF STUDY >
- Pharmacokinetic Study on ADME (absorption, distribution, metabolism, excretion) course
RESULTS >
- Peak plasma concentration occurred at about 2 hours after oral administration
- Good distribution to tissues and organs
- High excretion in feces
- Absolute cumulative contribution ratio increased slowly from 48.3, 57.3, to 65.3% for 1, 2, and 3 weeks of repetitive oral administration and decreased rapidly for the 1st week and then slowly for 1 to 2 weeks of recovery period after the termination of administration
Anti-angiogenesis effect of As4O6
EXPERIMENT
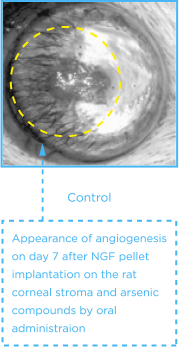
CORNEA OF RATS
(Source; J Vet Med Sci. 66:1091-5, 2004)
CASE REVIEW

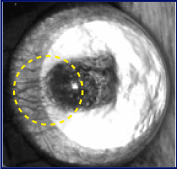
As2O3(Arsenic trioxide )
As4O6 (Arsenic hexaoxide)
RESULTS
The number, length, clock hour of neovascularization and areas of the vessels in As4O6 group showed more significant inhibition than those of control and As2O3 groups from day 5
EXPERIMENT
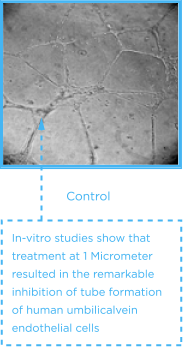
Tube forming assay With HUVEC (Vascular Endothelial Cells)
(Source; Int J Oncol; 22(6):1271-6, 2003)
CASE REVIEW
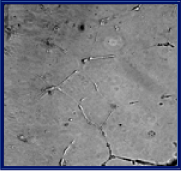
1 µM → As2O3
1 µM → As4O6
RESULTS
It was found that antiangiogenesis effect of vascular endothelial cells administered with As4O6 were better than that of As2O3.
EXPERIMENT
Experimental Melanoma Lung Metastasis Study
CASE REVIEW
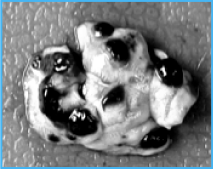
Control
Inject B16F10 melanoma cells into
the tail vein of C57BL/6 mice
(7.5×104 cells/animal)
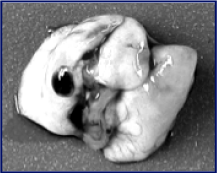
As4O6 Treated
Oral administration of As4O6
after the tumor inoculation and
the number of lung nodules was
counted 22 days later
(9 mice were used for the control
group and 10 mice for the
treatment group)
RESULTS
* The number of lung nodules
Control group: 93.1 +-6.3
As4O6 Treated: 39.4 +-6.3
As4O6 Treated represents 54% reduction in the tumor number
LD50
Male SD rat: 50mg/kg
Female SD rat: 50mg/kg
Male mice: 60mg/kg
Female mice: 60mg/kg
It was identified that the cancer therapeutic effect of CM-7919 was remarkably enhanced in combination therapy with radiation and chemotherapy
* Source: Korea Cancer Center Hospital, Busan Paik Hospital
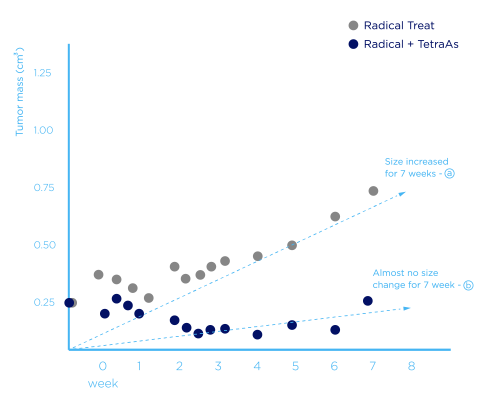
* Observed the progress of tumor for 7 weeks after 1 week radiation
therapy only ⓐ and 1 week combination therapy of radiation
and CM-7919 ⓑ
ⓐ The observed period, the size of tumor increased again
ⓑ Up to 7 weeks, the size of tumor was reduced or changed little (combination cancer therapy effect)
<Journal> Tetra-arsenic oxide (TetraAs) enhances radiation sensitivity of solid tumors by anti-vascular effect (Cancer Letters, 2009.01.20)
* Source: School of medicine, Catholic University of Korea
![]() Enhanced therapeutic effect :
Enhanced therapeutic effect :
<Journal> Anti-proliferative Effect of Tetra-arsenic Oxide (TetraAs) in Human Gastric Cancer Cells in Vitro (Journal of Pharmaceutical Investigation, 2007)
